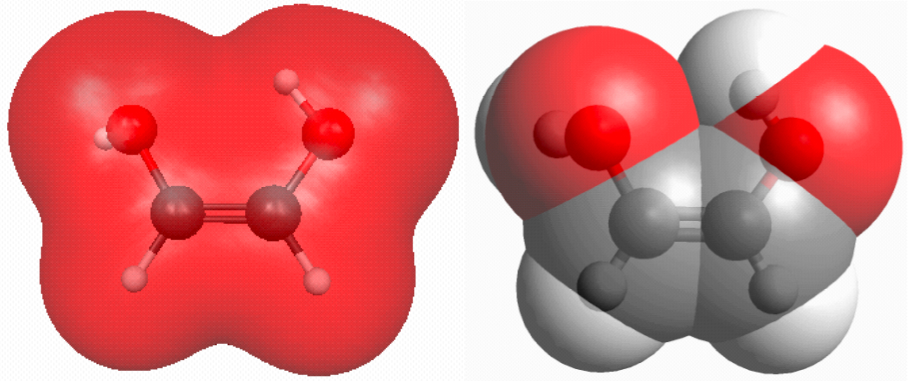
Figure 3.3. (a) RHF/6-31G(d) 0.002 au isodensity surface and (b) van der Waals surface of cis-HO(H)C=C(H)OH (click on the picture for a bigger version).
From Molecular Modeling Basics CRC Press, May 2010.
From Molecular Modeling Basics CRC Press, May 2010.
Looking at the electron density or the van der Waals surfaces one would expect steric strain in cis-HO(H)C=C(H)OH. (See this post on how to make such pictures and this post on how to make an interactive version such as the one shown below.)
However, contrary to CH3(H)C=C(H)CH3, the cis isomer is more stable than the trans isomer. This is of course due to the electrostatic attraction of the O and H atom in the hydrogen bond. Here the electron density tells only part of the story because this attraction involves the nuclei as well. Here an electrostatic potential map is more useful. How to make such plots will be the subject of future posts.
Figure 3.3. The RHF/6-31G(d) electron density of cis-HO(H)C=C(H)OH.
Click on the picture for an interactive version
Click on the picture for an interactive version




No comments:
Post a Comment