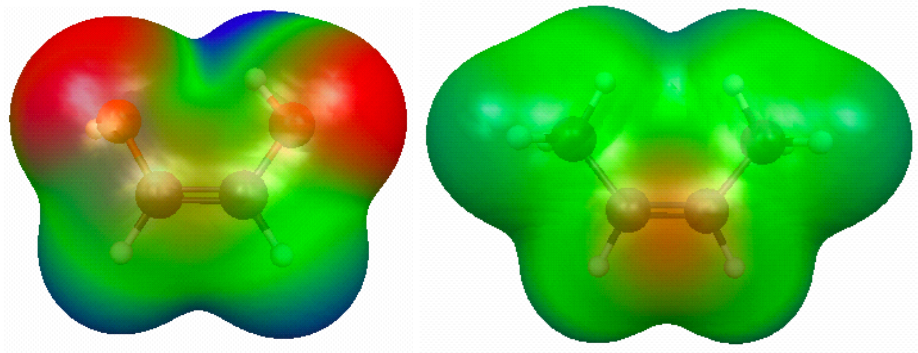
Figure 3.5. (a) RHF/6-31G(d) 0.002 au isodensity surface with superimposed electrostatic potential for (a) cis-HO(H)C=C(H)OH and (b) cis-CH3(H)C=C(H)CH3 and. In both cases, the maximum potential value is 0.05 au. (click on the picture for a bigger version).
From Molecular Modeling Basics CRC Press, May 2010.
From Molecular Modeling Basics CRC Press, May 2010.
This figure shows how the electrostatic potential superimposed on the 0.002 au isodensity surface can be used to rationalize why cis-HO(H)C=C(H)OH is more stable than trans-HO(H)C=C(H)OH, while the opposite is true for CH3(H)C=C(H)CH3.
The figure clearly shows the difference in polarity between hydroxyl and methyl groups. For the OH substitutent case the positive (O)H atom is close to the negative O(H) atom in the cis isomer. The CH3 group is non-polar and larger and the overlapping density indicates steric strain.
The figure is made with MacMolPlt as described in a previous post. Below is an interactive version made with Jmol (as described in a previous post). You'll notice that the color scheme is somewhat different, because Jmol maps the electrostatic potential value to a color in a different way than MacMolPlt. However, the general conclusion drawn from both programs is clearly the same.
Figure 3.5. (a) RHF/6-31G(d) 0.002 au isodensity surface with superimposed electrostatic potential for (a) cis-HO(H)C=C(H)OH and (b) cis-CH3(H)C=C(H)CH3 and.
Click on the picture for an interactive version
Click on the picture for an interactive version




No comments:
Post a Comment